 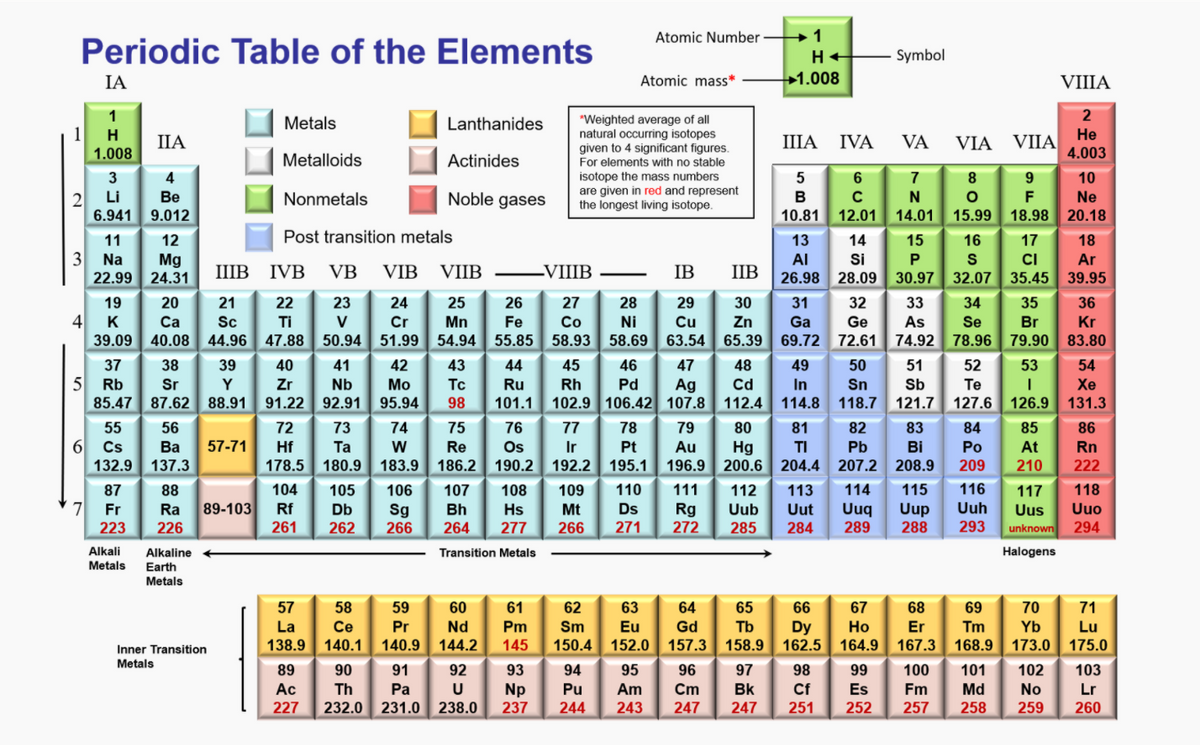
The gas with the smaller molar mass will have a larger fraction of “fast molecules” and a smaller fraction of “slow molecules”, no matter where we choose the cutoff between “fast” and “slow”. The molar mass of an element is determined by the atomic weight of that element, which can be found on the periodic table. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10-19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. If the temperature is constant, the velocity distribution is inversely dependent on the molar mass the smaller the molar mass, the faster the molecules.Ĭ) The fraction of CH 4 molecules with velocities greater than 500 m/sec is larger. Chlorine is a chemical element with atomic number 17 which means there are 17 protons in its nucleus. Which substance has a molar mass of 74. To two decimal places, the formula mass of NaCl is 58.44 amu. :max_bytes(150000):strip_icc()/PeriodicTableSigFigBW-58b5c7f25f9b586046cae098.png)
The number of grams is determined by adding up the atomic masses of the individual elements on the periodic table. Multiply the number of atoms by the atomic weight of each element found in steps 1 and 2 to get the mass of each element in C2H5Cl: Molar Mass (g/mol) C (Carbon) 2 × 12.0107 24.0214. This formula mass is the sum of the atomic masses of one sodium atom and one chlorine atom, which we find from the periodic table here, we use the masses to two decimal places: Na: 22.99 amu. Visualize trends, 3D orbitals, isotopes, and mix compounds. This results in the units of g/mol for molar mass. Interactive periodic table showing names, electrons, and oxidation states. 
The rms velocity depends on both the molar mass and the temperature, so it is easiest to just calculate the rms velocity for each gas, v rms for N 2 is 515 m/s and for O 2 is 521 m/s.ī) Hydrogen is larger. Molar mass is the mass of one mole, in grams, of a given substance. (“KE” means kinetic energy)Ī) The rms velocity of nitrogen at 25✬ or the rms velocity of oxygen at 75✬.ī) The average velocity of hydrogen or the average velocity of helium at the same temperature.Ĭ) The fraction of \(CH_4\) molecules with velocities greater than 500 m/sec, or the fraction of \(NH_3\) molecules with velocities greater than 500 m/sec at the same temperature.Ī) O xygen at 75✬ is larger. If they are equal, say so and explain how you can tell. \): Which is larger?įor each of the following pairs of quantities, tell which one is larger and explain your answer. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
